Atmosphere of Earth

The atmosphere of Earth is a layer of gases surrounding the planet Earth that is retained by Earth's gravity. The atmosphere protects life on Earth by absorbing ultraviolet solar radiation, warming the surface through heat retention (greenhouse effect), and reducing temperature extremes between day and night. Dry air contains roughly (by volume) 78.09% nitrogen, 20.95% oxygen, 0.93% argon, 0.039% carbon dioxide, and small amounts of other gases. Air also contains a variable amount of water vapor, on average around 1%.
The atmosphere has a mass of about 5 × 1018 kg, three quarters of which is within about 11 km (6.8 mi; 36,000 ft) of the surface. The atmosphere becomes thinner and thinner with increasing altitude, with no definite boundary between the atmosphere and outer space. An altitude of 120 km (75 mi) is where atmospheric effects become noticeable during atmospheric reentry of spacecraft. The Kármán line, at 100 km (62 mi), also is often regarded as the boundary between atmosphere and outer space.
Contents |
Composition
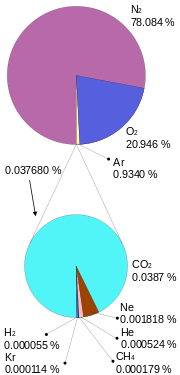

Air is mainly composed of nitrogen, oxygen, and argon, which together constitute the major gases of the atmosphere. The remaining gases are often referred to as trace gases,[1] among which are the greenhouse gases such as water vapor, carbon dioxide, methane, nitrous oxide, and ozone. Filtered air includes trace amounts of many other chemical compounds. Many natural substances may be present in tiny amounts in an unfiltered air sample, including dust, pollen and spores, sea spray, and volcanic ash. Various industrial pollutants also may be present, such as chlorine (elementary or in compounds), fluorine compounds, elemental mercury, and sulfur compounds such as sulfur dioxide [SO2].
| ppmv: parts per million by volume (note: volume fraction is equal to mole fraction for ideal gas only, see Gas Volume) | |
| Gas | Volume |
|---|---|
| Nitrogen (N2) | 780,840 ppmv (78.084%) |
| Oxygen (O2) | 209,460 ppmv (20.946%) |
| Argon (Ar) | 9,340 ppmv (0.9340%) |
| Carbon dioxide (CO2) | 390 ppmv (0.039%) |
| Neon (Ne) | 18.18 ppmv (0.001818%) |
| Helium (He) | 5.24 ppmv (0.000524%) |
| Methane (CH4) | 1.79 ppmv (0.000179%) |
| Krypton (Kr) | 1.14 ppmv (0.000114%) |
| Hydrogen (H2) | 0.55 ppmv (0.000055%) |
| Nitrous oxide (N2O) | 0.3 ppmv (0.00003%) |
| Carbon monoxide (CO) | 0.1 ppmv (0.00001%) |
| Xenon (Xe) | 0.09 ppmv (9 × 10−6%) |
| Ozone (O3) | 0.0 to 0.07 ppmv (0% to 7 × 10−6%) |
| Nitrogen dioxide (NO2) | 0.02 ppmv (2 × 10−6%) |
| Iodine (I) | 0.01 ppmv (1 × 10−6%) |
| Ammonia (NH3) | trace |
| Not included in above dry atmosphere: | |
| Water vapor (H2O) | ~0.40% over full atmosphere, typically 1%-4% at surface |
Structure of the atmosphere
Principal layers

Earth's atmosphere can be divided into five main layers. These layers are mainly determined by whether temperature increases or decreases with altitude. From highest to lowest, these layers are:
- Exosphere
- The outermost layer of Earth's atmosphere extends from the exobase upward. Here the particles are so far apart that they can travel hundreds of kilometres without colliding with one another. Since the particles rarely collide, the atmosphere no longer behaves like a fluid. These free-moving particles follow ballistic trajectories and may migrate into and out of the magnetosphere or the solar wind. The exosphere is mainly composed of hydrogen and helium.
- Thermosphere
- Temperature increases with height in the thermosphere from the mesopause up to the thermopause, then is constant with height. The temperature of this layer can rise to 1,500 °C (2,730 °F), though the gas molecules are so far apart that temperature in the usual sense is not well defined. The International Space Station orbits in this layer, between 320 and 380 km (200 and 240 mi). The top of the thermosphere is the bottom of the exosphere, called the exobase. Its height varies with solar activity and ranges from about 350–800 km (220–500 mi; 1,100,000–2,600,000 ft).
- Mesosphere
- The mesosphere extends from the stratopause to 80–85 km (50–53 mi; 260,000–280,000 ft). It is the layer where most meteors burn up upon entering the atmosphere. Temperature decreases with height in the mesosphere. The mesopause, the temperature minimum that marks the top of the mesosphere, is the coldest place on Earth and has an average temperature around −85 °C (−121 °F; 188.2 K)[3]. Due to the cold temperature of the mesophere, water vapor is frozen, forming ice clouds (or Noctilucent clouds). A type of lightning referred to as either sprites or ELVES, form many miles above thunderclouds in the troposphere.
- Stratosphere
- The stratosphere extends from the tropopause to about 51 km (32 mi; 170,000 ft). Temperature increases with height, which restricts turbulence and mixing. The stratopause, which is the boundary between the stratosphere and mesosphere, typically is at 50 to 55 km (31 to 34 mi; 160,000 to 180,000 ft). The pressure here is 1/1000th sea level.
- Troposphere
- The troposphere begins at the surface and extends to between 7 km (23,000 ft) at the poles and 17 km (56,000 ft) at the equator, with some variation due to weather. The troposphere is mostly heated by transfer of energy from the surface, so on average the lowest part of the troposphere is warmest and temperature decreases with altitude. This promotes vertical mixing (hence the origin of its name in the Greek word "τροπή", trope, meaning turn or overturn). The troposphere contains roughly 80% of the mass of the atmosphere. The tropopause is the boundary between the troposphere and stratosphere.
Other layers
Within the five principal layers determined by temperature are several layers determined by other properties.
- The ozone layer is contained within the stratosphere. In this layer ozone concentrations are about 2 to 8 parts per million, which is much higher than in the lower atmosphere but still very small compared to the main components of the atmosphere. It is mainly located in the lower portion of the stratosphere from about 15–35 km (9.3–22 mi; 49,000–110,000 ft), though the thickness varies seasonally and geographically. About 90% of the ozone in our atmosphere is contained in the stratosphere.
- The ionosphere, the part of the atmosphere that is ionized by solar radiation, stretches from 50 to 1,000 km (31 to 620 mi; 160,000 to 3,300,000 ft) and typically overlaps both the exosphere and the thermosphere. It forms the inner edge of the magnetosphere. It has practical importance because it influences, for example, radio propagation on the Earth. It is responsible for auroras.
- The homosphere and heterosphere are defined by whether the atmospheric gases are well mixed. In the homosphere the chemical composition of the atmosphere does not depend on molecular weight because the gases are mixed by turbulence.[4] The homosphere includes the troposphere, stratosphere, and mesosphere. Above the turbopause at about 100 km (62 mi; 330,000 ft) (essentially corresponding to the mesopause), the composition varies with altitude. This is because the distance that particles can move without colliding with one another is large compared with the size of motions that cause mixing. This allows the gases to stratify by molecular weight, with the heavier ones such as oxygen and nitrogen present only near the bottom of the heterosphere. The upper part of the heterosphere is composed almost completely of hydrogen, the lightest element.
- The planetary boundary layer is the part of the troposphere that is nearest the Earth's surface and is directly affected by it, mainly through turbulent diffusion. During the day the planetary boundary layer usually is well-mixed, while at night it becomes stably stratified with weak or intermittent mixing. The depth of the planetary boundary layer ranges from as little as about 100 m on clear, calm nights to 3000 m or more during the afternoon in dry regions.
The average temperature of the atmosphere at the surface of Earth is 14 °C (57 °F; 287 K)[5] or 15 °C (59 °F; 288 K)[6], depending on the reference.[7] [8][9]
Physical properties
Pressure and thickness
The average atmospheric pressure at sea level is about 1 atmosphere (atm) = 101.3 kPa (kilopascals) = 14.7 psi (pounds per square inch) = 760 torr = 29.9 inches of mercury (symbol Hg). Total atmospheric mass is 5.1480×1018 kg (1.135×1019 lb),[10] about 2.5% less than would be inferred naively from the average sea level pressure and the Earth's area of 51007.2 megahectares, this defect having been displaced by the Earth's mountainous terrain. Atmospheric pressure is the total weight of the air above unit area at the point where the pressure is measured. Thus air pressure varies with location and time, because the amount of air above the Earth's surface varies.
If atmospheric density were to remain constant with height the atmosphere would terminate abruptly at 8.50 km (27,900 ft). Instead, density decreases with height, dropping by 50% at an altitude of about 5.6 km (18,000 ft). As a result the pressure decrease is approximately exponential with height, so that pressure decreases by a factor of two approximately every 5.6 km (18,000 ft) and by a factor of e = 2.718… approximately every 7.64 km (25,100 ft), the latter being the average scale height of Earth's atmosphere below 70 km (43 mi; 230,000 ft). However, because of changes in temperature, average molecular weight, and gravity throughout the atmospheric column, the dependence of atmospheric pressure on altitude is modeled by separate equations for each of the layers listed above. Even in the exosphere, the atmosphere is still present. This can be seen by the effects of atmospheric drag on satellites.
In summary, the equations of pressure by altitude in the above references can be used directly to estimate atmospheric thickness. However, the following published data are given for reference:[11]
- 50% of the atmosphere by mass is below an altitude of 5.6 km (18,000 ft).
- 90% of the atmosphere by mass is below an altitude of 16 km (52,000 ft). The common altitude of commercial airliners is about 10 km (33,000 ft) and Mt. Everest's summit is 8,848 m (29,029 ft) above sea level.
- 99.99997% of the atmosphere by mass is below 100 km (62 mi; 330,000 ft), although in the rarefied region above this there are auroras and other atmospheric effects. The highest X-15 plane flight in 1963 reached an altitude of 354,300 ft (108.0 km).
Density and mass

The density of air at sea level is about 1.2 kg/m3 (1.2 g/L). Density is not measured directly but is calculated from measurements of temperature, pressure and humidity using the equation of state for air (a form of the ideal gas law). Atmospheric density decreases as the altitude increases. This variation can be approximately modeled using the barometric formula. More sophisticated models are used to predict orbital decay of satellites.
The average mass of the atmosphere is about 5 quadrillion (5 × 1015) tonnes or 1/1,200,000 the mass of Earth. According to the National Center for Atmospheric Research, "The total mean mass of the atmosphere is 5.1480 × 1018 kg with an annual range due to water vapor of 1.2 or 1.5 × 1015 kg depending on whether surface pressure or water vapor data are used; somewhat smaller than the previous estimate. The mean mass of water vapor is estimated as 1.27 × 1016 kg and the dry air mass as 5.1352 ±0.0003 × 1018 kg."
Optical properties
Solar radiation (or sunlight) is the energy the Earth receives from the Sun. The Earth also emits radiation back into space, but at longer wavelengths that we cannot see. Part of the incoming and emitted radiation is absorbed or reflected by the atmosphere.
Scattering
When light passes through our atmosphere, photons interact with it through scattering. If the light does not interact with the atmosphere, it is called direct radiation and is what you see if you were to look directly at the Sun. Indirect radiation is light that has been scattered in the atmosphere. For example, on an overcast day when you cannot see your shadow there is no direct radiation reaching you, it has all been scattered. As another example, due to a phenomenon called Rayleigh scattering, shorter (blue) wavelengths scatter more easily than longer (red) wavelengths. This is why the sky looks blue, you are seeing scattered blue light. This is also why sunsets are red. Because the Sun is close to the horizon, the Sun's rays pass through more atmosphere than normal to reach your eye. Much of the blue light has been scattered out, leaving the red light in a sunset.
Absorption
Different molecules absorb different wavelengths of radiation. For example, O2 and O3 absorb almost all wavelengths shorter than 300 nanometers. Water (H2O) absorbs many wavelengths above 700 nm. When a molecule absorbs a photon, it increases the energy of the molecule. We can think of this as heating the atmosphere, but the atmosphere also cools by emitting radiation, as discussed below.
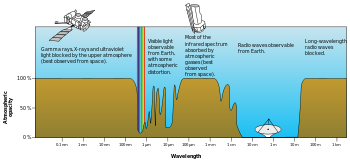
The combined absorption spectra of the gases in the atmosphere leave "windows" of low opacity, allowing the transmission of only certain bands of light. The optical window runs from around 300 nm (ultraviolet-C) up into the range humans can see, the visible spectrum (commonly called light), at roughly 400–700 nm and continues to the infrared to around 1100 nm. There are also infrared and radio windows that transmit some infrared and radio waves at longer wavelengths. For example, the radio window runs from about one centimeter to about eleven-meter waves.
Emission
Emission is the opposite of absorption, it is when an object emits radiation. Objects tend to emit amounts and wavelengths of radiation depending on their "black body" emission curves, therefore hotter objects tend to emit more radiation, with shorter wavelengths. Colder objects emit less radiation, with longer wavelengths. For example, the Sun is approximately 6,000 K (5,730 °C; 10,340 °F), its radiation peaks near 500 nm, and is visible to the human eye. The Earth is approximately 290 K (17 °C; 62 °F), so its radiation peaks near 10,000 nm, and is much too long to be visible to humans.
Because of its temperature, the atmosphere emits infrared radiation. For example, on clear nights the Earth's surface cools down faster than on cloudy nights. This is because clouds (H2O) are strong absorbers and emitters of infrared radiation. This is also why it becomes colder at night at higher elevations. The atmosphere acts as a "blanket" to limit the amount of radiation the Earth loses into space.
The greenhouse effect is directly related to this absorption and emission (or "blanket") effect. Some chemicals in the atmosphere absorb and emit infrared radiation, but do not interact with sunlight in the visible spectrum. Common examples of these chemicals are CO2 and H2O. If there are too much of these greenhouse gases, sunlight heats the Earth's surface, but the gases block the infrared radiation from exiting back to space. This imbalance causes the Earth to warm, and thus climate change.
Refractive index
The refractive index of air is close to, but just greater than 1. Systematic variations in refractive index can lead to the bending of light rays over long optical paths. One example is that, under some circumstances, observers onboard ships can see other vessels just over the horizon because light is refracted in the same direction as the curvature of the Earth's surface.
The refractive index of air depends on temperature, giving rise to refraction effects when the temperature gradient is large. An example of such effects is the mirage.
- See also: Scintillation (astronomy)
Circulation

Atmospheric circulation is the large-scale movement of air, and the means (with ocean circulation) by which heat is distributed around the Earth. The large-scale structure of the atmospheric circulation varies from year to year, but the basic structure remains fairly constant as it is determined by the Earth's rotation rate and the difference in solar radiation between the equator and poles.
Evolution of Earth's atmosphere
Second atmosphere
Water related sediments have been found dating from as early as 3.8 billion years ago.[12] About 3.4 billion years ago, nitrogen was the major part of the then stable "second atmosphere." An influence of life has to be taken into account rather soon in the history of the atmosphere, since hints of early life forms are to be found as early as 3.5 billion years ago.[13] The fact that this is not perfectly in line with the - compared to today 30% lower - solar radiance of the early Sun has been described as the "Faint young Sun paradox".
The geological record however shows a continually relatively warm surface during the complete early temperature record of the Earth with the exception of one cold glacial phase about 2.4 billion years ago. In the late Archaean era an oxygen-containing atmosphere began to develop, apparently from photosynthesizing algae which have been found as stromatolite fossils from 2.7 billion years ago. The early basic carbon isotopy (isotope ratio proportions) is very much in line with what is found today,[14] suggesting that the fundamental features of the carbon cycle were established as early as 4 billion years ago.
Third atmosphere

The accretion of continents about 3.5 billion years ago[15] added plate tectonics, constantly rearranging the continents and also shaping long-term climate evolution by allowing the transfer of carbon dioxide to large land-based carbonate storages. Free oxygen did not exist until about 1.7 billion years ago and this can be seen with the development of the red beds and the end of the banded iron formations. This signifies a shift from a reducing atmosphere to an oxidising atmosphere. O2 showed major ups and downs until reaching a steady state of more than 15%.[16] The following time span was the Phanerozoic era, during which oxygen-breathing metazoan life forms began to appear.
Currently, anthropogenic greenhouse gases are increasing in the atmosphere. According to the Intergovernmental Panel on Climate Change, this increase is the main cause of global warming.[17]
Air pollution
Air pollution is the human introduction of chemicals, particulate matter, or biological materials that cause harm or discomfort to organisms into the atmosphere.[18] Stratospheric ozone depletion is believed to be caused by air pollution (chiefly from chlorofluorocarbons).
See also
- Aerial perspective
- Air glow
- Airshed
- Atmosphere (for information on atmospheres in general)
- Atmospheric dispersion modeling
- Atmospheric electricity
- Atmospheric models
- Atmospheric Radiation Measurement (ARM) (in the U.S.)
- Atmospheric stratification
- Aviation
- Biosphere
- Carbon dioxide in the Earth's atmosphere
- Compressed air
- Global dimming
- Historical temperature record
- Hydrosphere
- Intergovernmental Panel on Climate Change
- Kyoto Protocol
- Lithosphere
- Ozone layer
- Standard Dry Air
- US Standard Atmosphere
- Warm period
- Water vapor in Earth's atmosphere
References
- ↑ http://www.ace.mmu.ac.uk/eae/Atmosphere/Older/Trace_Gases.html
- ↑ Source for figures: Carbon dioxide, NOAA Earth System Research Laboratory, (updated 2010.06). Methane, IPCC TAR table 6.1, (updated to 1998). The NASA total was 17 ppmv over 100%, and CO2 was increased here by 15 ppmv. To normalize, N2 should be reduced by about 25 ppmv and O2 by about 7 ppmv.
- ↑ States, Robert J.; Gardner, Chester S. (January 2000), "Thermal Structure of the Mesopause Region (80–105 km) at 40°N Latitude. Part I: Seasonal Variations", Journal of the Atmospheric Sciences 2000 57: 66-77, http://journals.ametsoc.org/doi/full/10.1175/1520-0469%282000%29057%3C0066%3ATSOTMR%3E2.0.CO%3B2
- ↑ homosphere—AMS Glossary
- ↑ "Earth's Atmosphere". http://www.bambooweb.com/articles/e/a/Earth's_atmosphere.html.
- ↑ NASA - Earth Fact Sheet
- ↑ "Global Surface Temperature Anomalies". http://www.ncdc.noaa.gov/oa/climate/research/anomalies/index.php.
- ↑ "Earth's Radiation Balance and Oceanic Heat Fluxes". http://oceanworld.tamu.edu/resources/oceanography-book/radiationbalance.htm.
- ↑ "Coupled Model Intercomparison Project Control Run". http://www-pcmdi.llnl.gov/projects/cmip/overview_ms/control_tseries.pdf.
- ↑ The Mass of the Atmosphere: A Constraint on Global Analyses
- ↑ Lutgens, Frederick K. and Edward J. Tarbuck (1995) The Atmosphere, Prentice Hall, 6th ed., pp14-17, ISBN 0-13-350612-6
- ↑ B. Windley: The Evolving Continents. Wiley Press, New York 1984
- ↑ J. Schopf: Earth’s Earliest Biosphere: Its Origin and Evolution. Princeton University Press, Princeton, N.J., 1983
- ↑ Celestial climate driver: a perspective from 4 billion years of the carbon cycle Geoscience Canada, March, 2005 by Jan Veizer
- ↑ Veizer in B. F. Windley (ed.), The Early History of the Earth, John Wiley and Sons, London, p. 569., 1976
- ↑ Christopher R. Scotese, Back to Earth History : Summary Chart for the Precambrian, Paleomar Project
- ↑ "Summary for Policymakers" (PDF). Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Intergovernmental Panel on Climate Change. 5 February 2007. http://www.ipcc.ch/pdf/assessment-report/ar4/wg1/ar4-wg1-spm.pdf.
- ↑ Starting from [1] Pollution - Definition from the Merriam-Webster Online Dictionary
External links
- NASA atmosphere models
- NASA's Earth Fact Sheet
- American Geophysical Union: Atmospheric Sciences
- Stuff in the Air Find out what the atmosphere contains.
- Layers of the Atmosphere
- Answers to several questions of curious kids related to Air and Atmosphere
- The AMS Glossary of Meteorology
- Paul Crutzen Interview Free video of Paul Crutzen Nobel Laureate for his work on decomposition of ozone talking to Harry Kroto Nobel Laureate by the Vega Science Trust.
- Slides describing the Earth's modern atmosphere
- Air Property
|
|||||||||||
|
|||||||||||||
|
||||||||||||||||||||